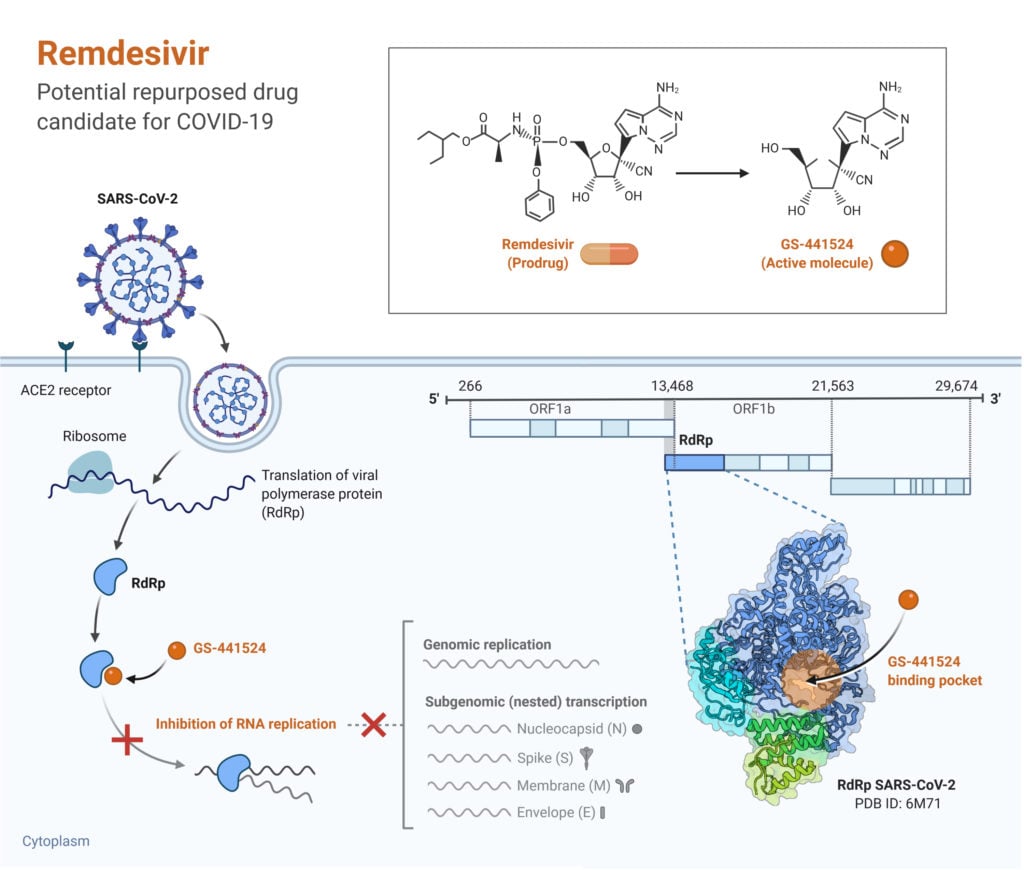
An NIR laser triggers the photothermally induced structural disruption of the nano-assembly, releasing the drug at the targeted sites. The binding of β-Glucan conjugated HTNs to the dectin-1 receptor present on macrophages increases the free radical production and cellular uptake of HTNs. Hence, host-targeted nano-assemblies (HTNs) were fabricated by conjugating host targeting ligands (β-Glucan) onto the nano-assembly. Mtb can alter immune defense mechanisms exerted by the host macrophage. The antibacterial activity of the final nano-assembly against Mycobacterium smegmatis (Msmeg) was 20 folds more efficacious than the free drug equivalent. Upon NIR laser irradiation GNRs convert the photon energy of the laser to localized heat, which melts the TSL, triggering the release of bedaquiline. NZX mediates the adhesion of the final nano-assembly onto the mycobacterial surface. The TSL layer is further conjugated to the mycobacteria-targeting peptide NZX. The core-shell nanoparticle is encapsulated within a thermo-sensitive liposome (TSL). The MS shell serves as a carrier for the anti-tuberculosis drug bedaquiline. The core of the nano-assembly is composed of GNRs coated with a mesoporous silica shell (MS). The protected delivery of antibiotics and their release in the proximity of the bacterial surface decreases off-target toxicity and drug dosage. The synergistic application of both therapies increases the efficacy of treatment. This NIR laser-activated nano-assembly delivers antibiotics to the site of infection and offers PTT. This thesis discusses the development of a combination therapeutic approach that kills intracellular bacteria in conjunction with photothermal and antibiotic therapy using gold nanorod (GNR) based nano-assembly. Gold-based nanomaterials have been widely used for biomedical applications such as photothermal therapy (PTT). Nanomaterials-based hybrid nano therapy is gaining attraction as a promising way to treat intracellular bacterial infections. Here, we review the currently developed PROTACs as the representative TPD molecules for cancer therapy and the N-degrons of the N-degron pathways as the potential TPD ligands. 
Among recent approaches to overcoming drug resistance to cancers, targeted protein degradation (TPD) such as proteolysis-targeting chimera (PROTAC) technology adopts a distinct mechanism of action by which a target protein is destroyed through the cellular proteolytic system, such as the ubiquitin–proteasome system or autophagy. However, long-term and high-dosage treatment of small inhibitors for cancer has produced other obstacles, such as resistance to inhibitors. The small molecule inhibitors for these target molecules greatly improved therapeutic efficacy and lowered the systemic toxicity in cancer therapies. Most target molecules are proteins such as kinases and kinase-associated receptors, which have enzymatic activities needed for the signaling cascades of cells. Extensive progress in understanding the molecular mechanisms of cancer growth and proliferation has led to the remarkable development of drugs that target cancer-driving molecules.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
